|
2/16/2024 0 Comments Nitrogen valence electrons 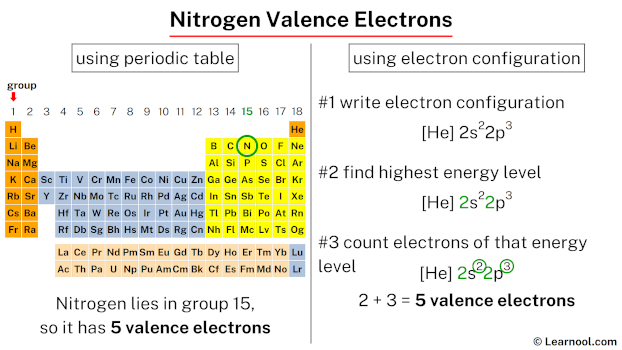
Here with our two hydrogens and a lone pair ofĮlectrons on the nitrogen. So in our drawing, nitrogen is surrounded by five valance electrons. Of electrons on the nitrogen, so that's four, and five. This would be one, two, three, and then we have a lone pair We have around nitrogen in our drawing? Let's count them up. Nitrogen gets one of the electrons and hydrogen gets the other. And same for this nitrogen-hydrogen bond. 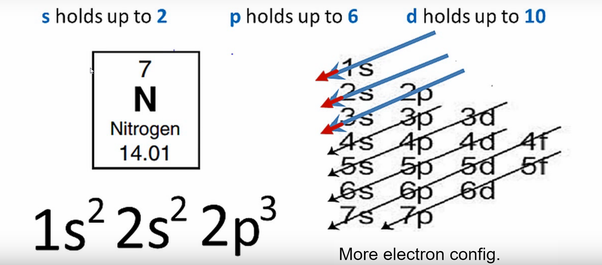
And from this bond on the right nitrogen gets one of those electrons and hydrogen gets the other. We know that from thisīond here on the left nitrogen gets one of those electrons. Here to the dot structure and let's look at these bonds. And from that we subtract the number of valence electrons that We know that nitrogen is supposed to have five valence electrons,īecause of its position on the periodic table. So formal charge is equal to the number of valence electrons that So I'm gonna draw in theĮlectrons in this bond so it's easier for us toĪssign a formal charge to the nitrogen. So let's assign a formalĬharge to the nitrogen in this molecule. Or another way of saying that, formal charge is equal to the number of valence electrons theĪtom is supposed to have minus the number of valence electrons that the atom actually has in the drawing. Number of valence electrons in the bonded atom. Number of valance electrons in the free atom minus the We'll assign formal charge to nitrogen, and just to remind you of the definition for formal charge, formal charge is equal to the
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
